Bryn Pharma presents data showing patient preference for Bi-dose Epinephrine Nasal Spray over EpiPen - NS Medical Devices

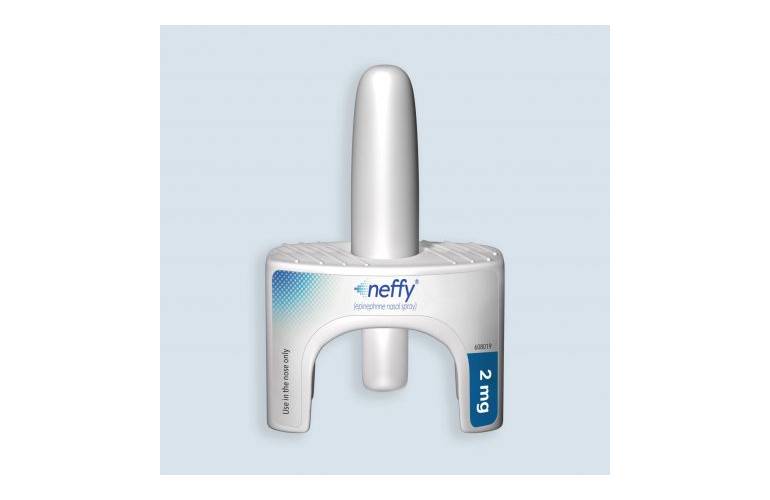
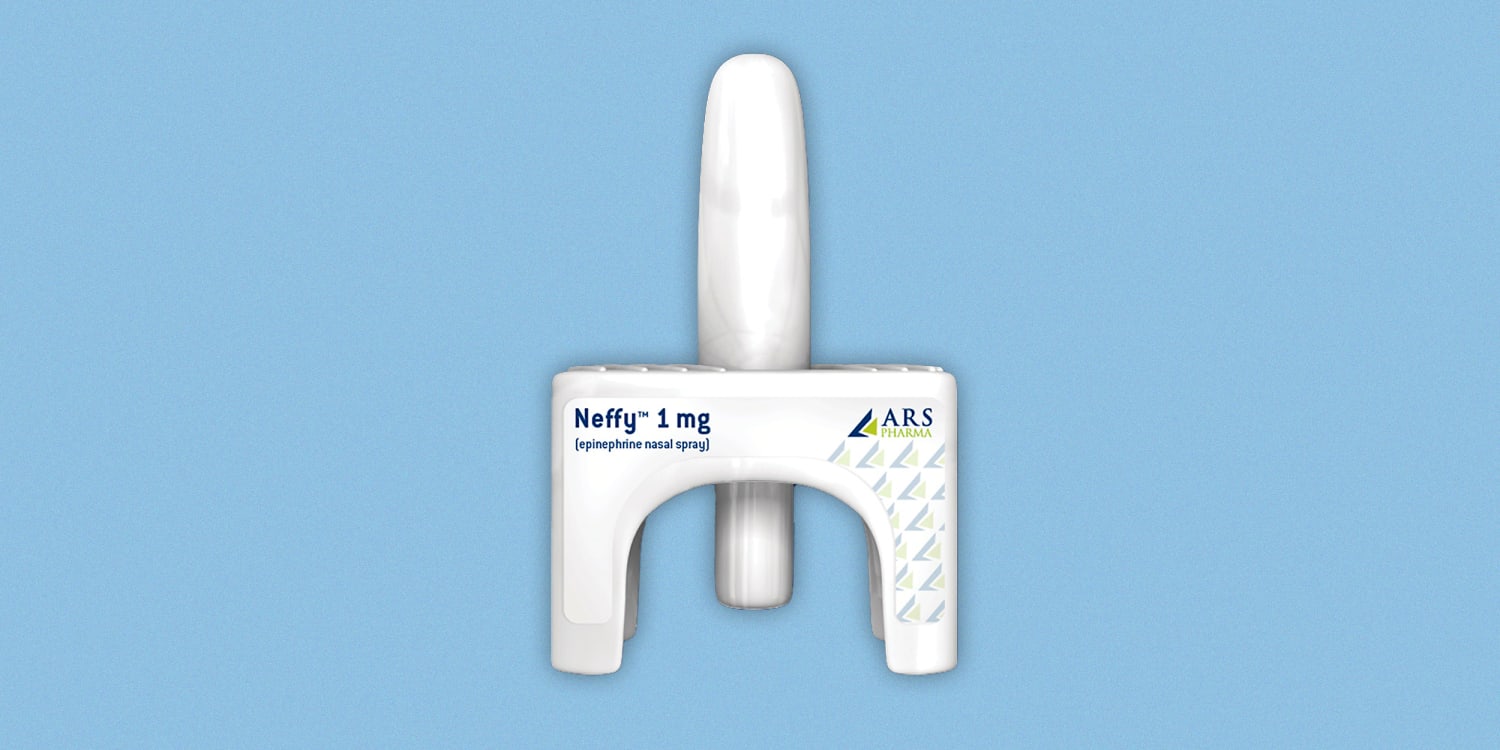
End Allergies Together - According to Bryn Pharma, a privately held pharmaceutical company dedicated to finding a better way for patients and caregivers to treat anaphylaxis, "Bryn has developed an epinephrine nasal

Statement on FDA Decision to Delay New Epinephrine Nasal Spray for Anaphylaxis | Allergy & Asthma Network
Intra-Nasal Epinephrine Takes Step Forward After Three Positive Pre-Clinical Studies | SnackSafely.com

Red Sneakers For Oakley - Wouldn't you love an epinephrine nasal spray could be an effective needle-free alternative for treating anaphylaxis? In a recent press release, The American Academy of Allergy, Asthma,

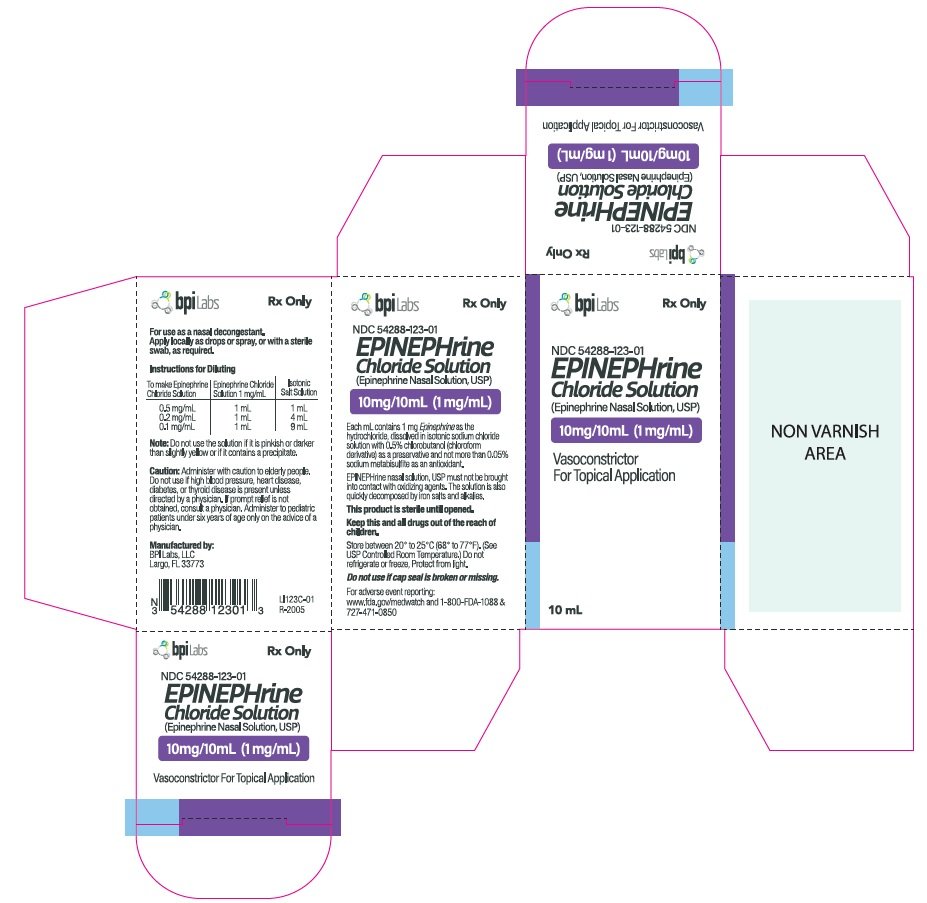
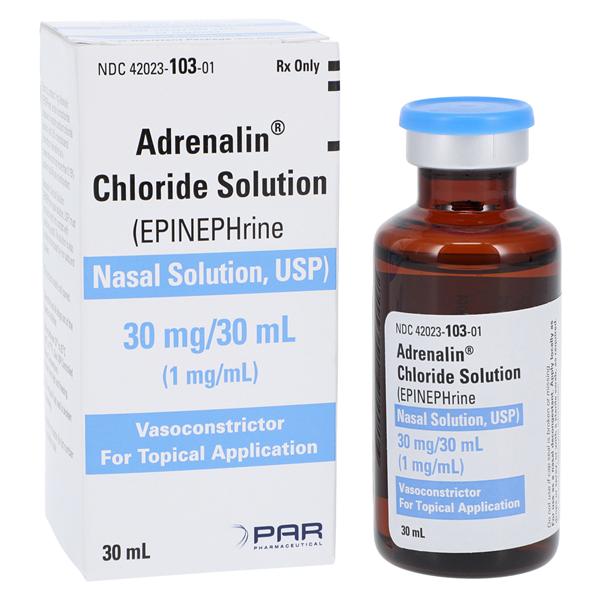
BPI Labs Epinephrine Chloride Solution (Epinephrine Nasal Solution) 10 — Mountainside Medical Equipment