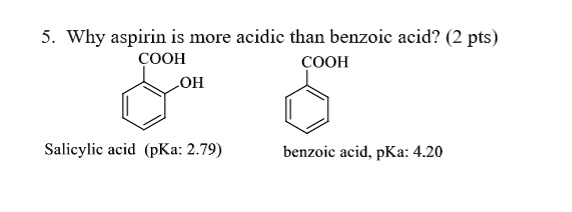
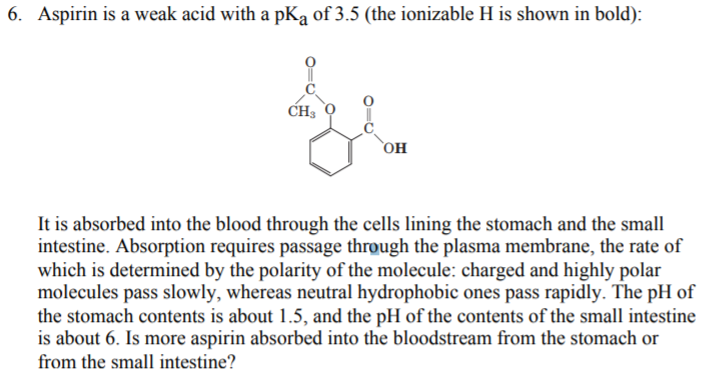
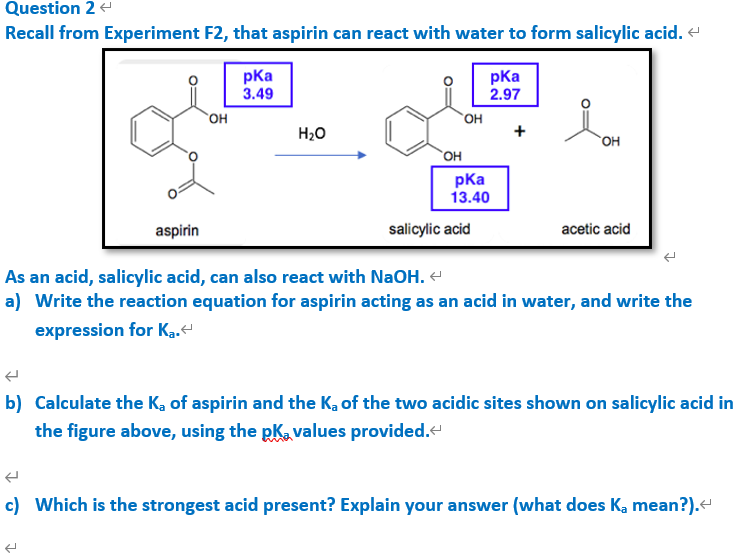
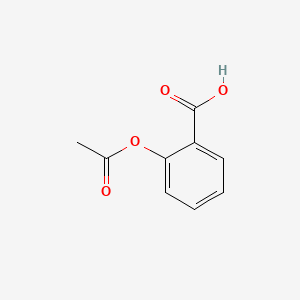
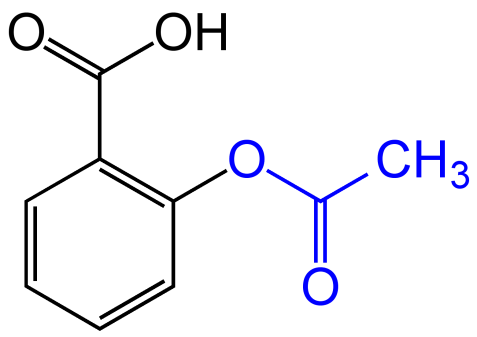
The pKa of acetylsalicylic acid (aspirin) is 3.5. The pH of gastric juice in the human stomach is about 2 - 3 and pH in the small intestine is about 8, aspirin will be:
The ${\\text{pKa}}$ of acetyl salicylic acid (aspirin) is $3.5$ . The ${\\text{pH}}$ of gastric juice in the human stomach is about $2 - 3$ and the ${\\text{pH}}$ in the small intestine is
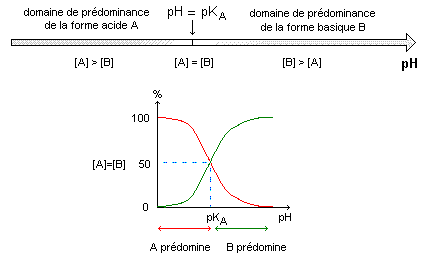
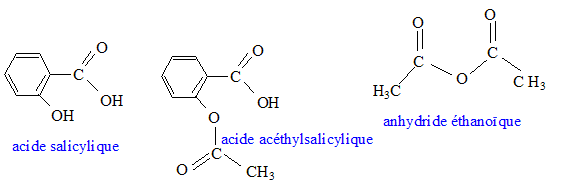
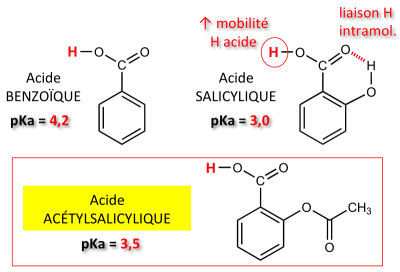
Chimie : structure ; propriétés de l'aspirine ; physique : étude d'oscillateurs ; optique géométrique et ondulatoire

ED3-Acide Base Redox - 1 UE1 CHIMIE EQUILIBRES ACIDO-BASIQUES ET REDOX EN SOLUTION AQUEUSE 1. - Studocu
LE CONSEIL DE LA PHARMACIENNE BACCALAURÉAT SÉRIE S Épreuve de PHYSIQUE CHIMIE Évaluation des Compétences Expérimentales So